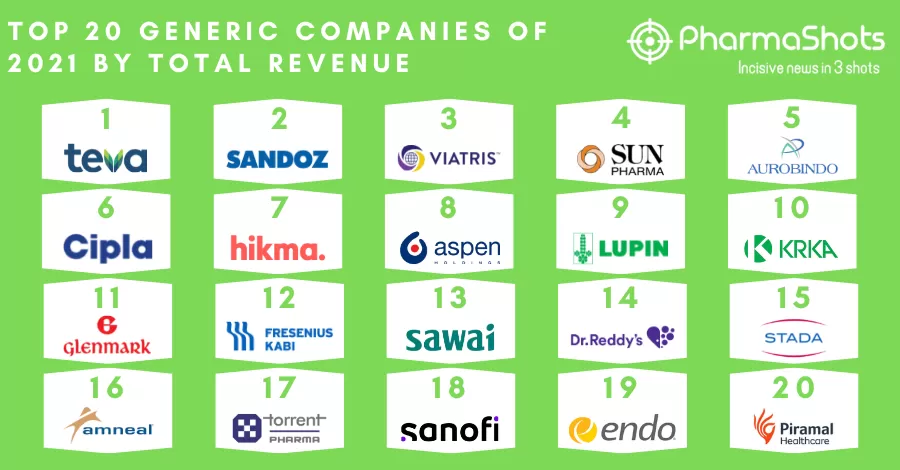
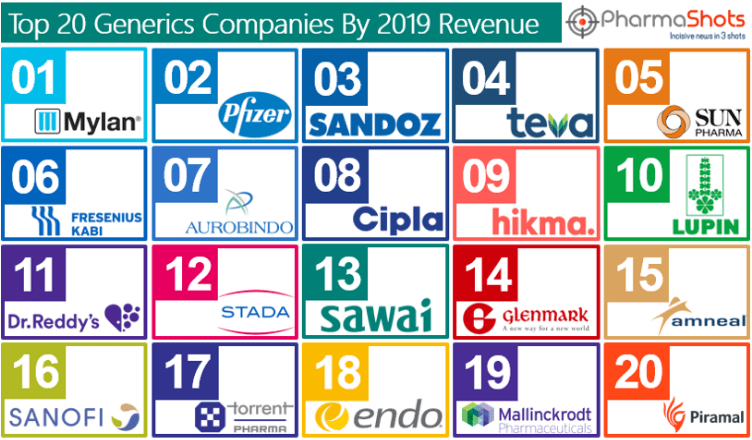
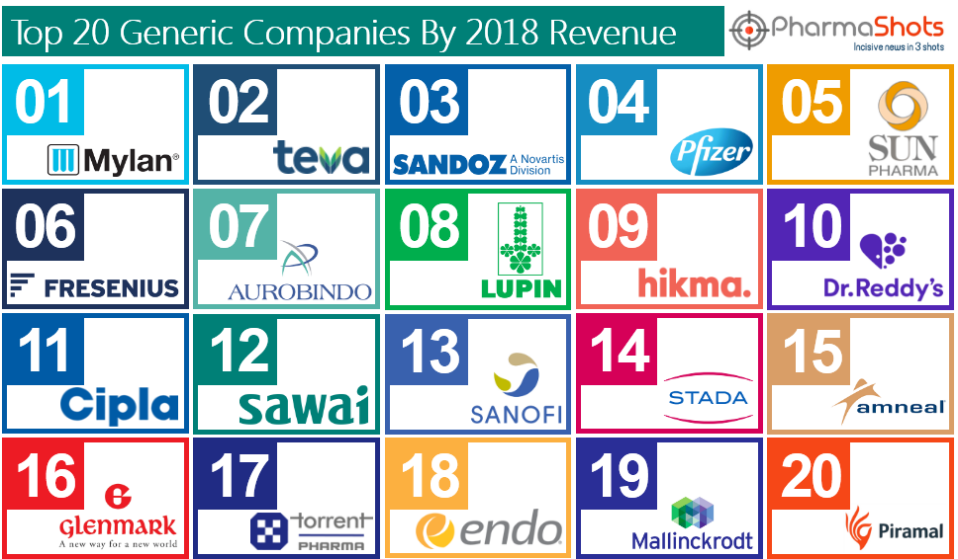
Aurobindo Pharma Subsidiary CuraTeQ Signs an Exclusive License Agreement with BioFactura to Commercialize BFI-751, a proposed biosimilar to Stelara
Shots:
- BioFactura will receive license fees of ~$33.5M across different milestones leading to commercialization, and ~43% profit sharing depending on the territory. The collaboration focuses on improving outcomes, QoL, and accessibility, and enhancing the patient experience with high-quality biosimilar medicines
- CuraTeQ to get an exclusive license right to commercialize BFI-751 in all major regulated markets incl. US, EU, UK, Canada, Australia & New Zealand, and other semi-regulated & emerging markets globally, also get the global manufacturing rights which will be produced at CuraTeQ facilities in Hyderabad, India
- CuraTeQ plans to file the biosimilar in India & emerging markets as early as 2024 with regulated market filings in 2026
Ref: Cision PR Web | Image: Biofactura
Related News:- BioFactura Initiates P-I Study of BFI-751 (biosimilar- ustekinumab)
PharmaShots! Your go-to media platform for customized news ranging for multiple indications. For more information connect with us at connect@pharmashots.com
Click here to read the full press release

Neha is a Senior Editor at PharmaShots. She is passionate and very enthusiastic about recent updates and developments in the life sciences and pharma industry. She covers Biopharma, MedTech, and Digital health segments along with different reports at PharmaShots. She can be contacted at connect@pharmashots.com.